Dedicated employees
Select Differentiated Technologies
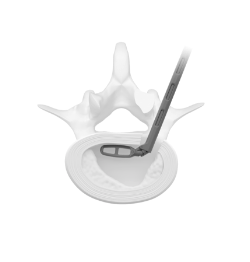
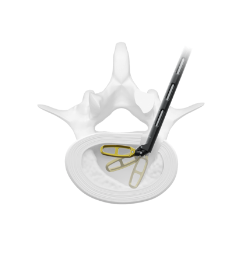
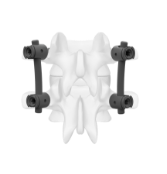
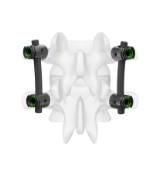
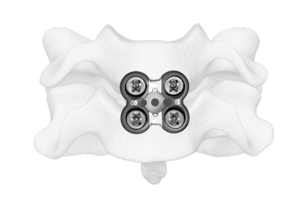
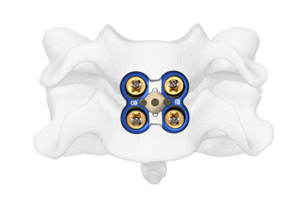
Metal-Free Fixation
Karma® Technology is a novel, metal-free system designed to stabilize the posterior column.

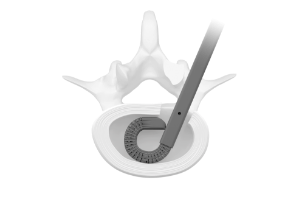
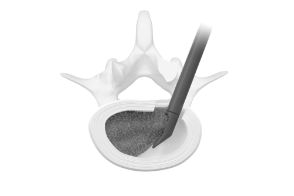
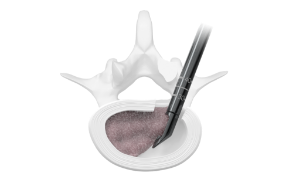
Enabling Access
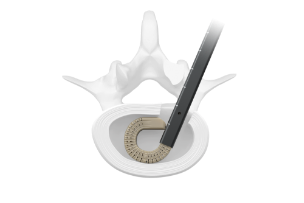
The Orbit® system consists of articulating and rotating discectomy instrumentation that uses a minimal posterior incision to achieve efficient disruption and removal of disc tissue.


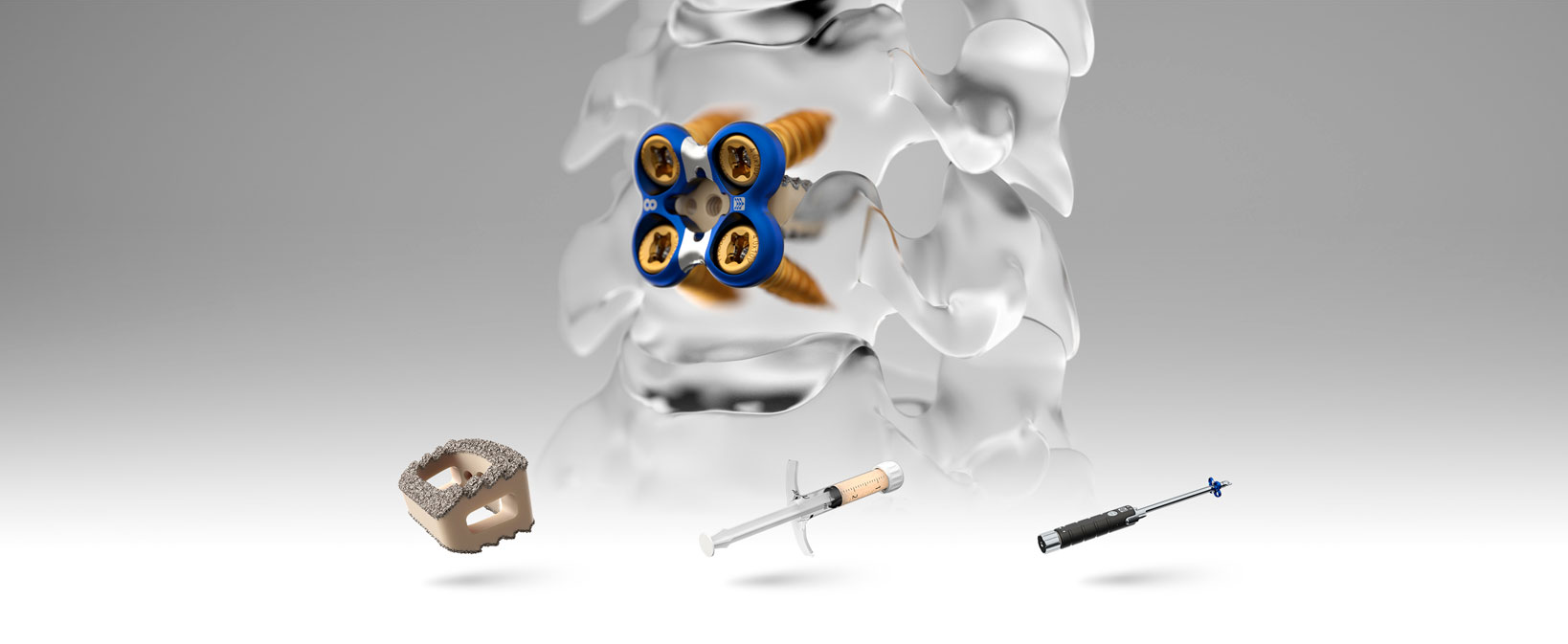
A comprehensive product suite for spinal surgery success
We designed our complete portfolio of advanced spinal implant technologies to help surgeons increase efficiency and reduce the stress of spine surgery on patients. Discover the Spinal Elements technology that fits your spine surgery needs.
Our company by the numbers
125 +
450 +
Issued U.S. & international patents
$ 1.5 M+
Donated to children's health charities
See what’s happening at Spinal Elements
Jan 09, 2024
View
Dec 20, 2023
View
Oct 13, 2023
View
Aug 03, 2023
View
Jun 20, 2023
View
Apr 19, 2023
View
Jul 20, 2022
View
Nov 08, 2021
View
Sep 22, 2021
View
Jun 08, 2021
View
Dec 21, 2020
View
Oct 30, 2020
View
Jun 17, 2020
View
Sep 20, 2019
View
Jan 14, 2019
View
Apr 13, 2017
View
Apr 26, 2016
View
Sep 14, 2015
View
Jan 21, 2015
View
Tomorrow’s spinal surgery solutions start here.
Learn more about how advanced, minimally invasive surgical solutions from Spinal Elements are transforming treatment and improving patient outcomes.